Identification And Design Of Novel Antimicrobial Peptides Targeting Mycobacterial Protein Kinase Pkn
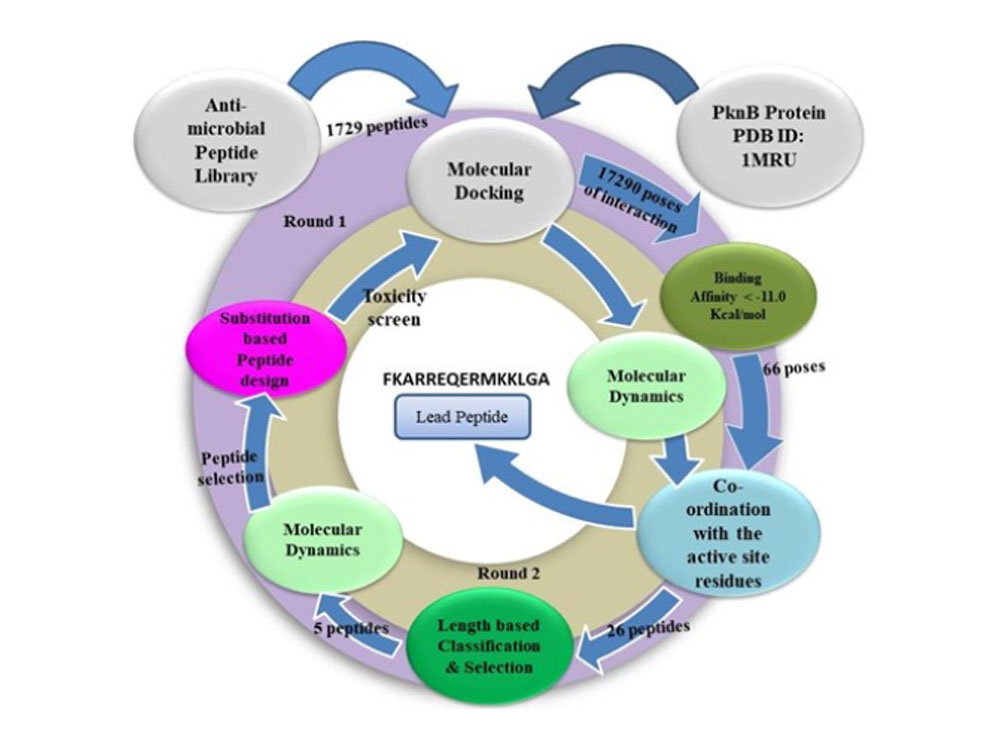
Antimicrobial peptides (AMPs) are increasingly favored over small molecule inhibitors due to their multifunctionality, ease of synthesis, and target specificity. Our recent study aimed to identify an AMP that inhibits PknB of M. tuberculosis, a serine/threonine protein kinase, by binding at its hinge region. A library of 5626 AMPs was prepared and categorized by length, and molecular docking identified peptides with consistent high-affinity binding to PknB’s active site. Twenty-six peptides were shortlisted and further grouped by length, with five chosen for molecular dynamic simulations in Gromacs based on binding affinity. Post-simulation analysis identified a 15-mer peptide as the most effective, leading to residue substitution to enhance interactions. Elastic Network Model (ENM) analysis further evaluated emphasizing the importance of peptide length and residue composition in developing effective peptide-based inhibitors. Article “Identification and Design of Novel Antimicrobial Peptides Targeting Mycobacterial Protein Kinase Pkn” published in The Protein Journal.

Leave a Reply
Want to join the discussion?Feel free to contribute!